
Some of the major advantages of CyTOF are the ability to run panel with 40–45 protein markers, barcoding for multiplexing samples, and the ability to cryopreserve samples after staining. Moreover, CyTOF has limitations such as low sample acquisition rate (around 1000 cells per second), and the destructive process prevents sorting of samples. CyTOF technology have theoretical capability to detect approximately 130 isotopes, but most of the published research articles used 40–45 protein markers (maximum panel size of 47 ) due to limitations in the availability of isotopes and signal spillover (minimal levels compared to fluorochrome spillover) that exist between isotopes. Advancements in multicolor flow cytometry and cytometry by time of flight (CyTOF) have enabled researchers to perform deep phenotyping of immune cells with sufficient sensitivity and resolution that is necessary to monitor the immune system of patients with impaired immune system such as cancer, autoimmune disorders, and so on and unravel the interaction between immune subsets. The panel has not been completely optimized but would rather act as a guide toward the development of a workflow for optimized multicolor immunophenotyping panel.įlow cytometry is the most frequently used high-throughput single-cells analysis technology that enables characterization of highly heterogenous samples such as blood and bone marrow. This panel can help to make a thorough interpretation of immune system, specifically when specimen quantity is low. Cells were analyzed on a 5-laser Cytek Aurora and data analysis was done using FlowJo. 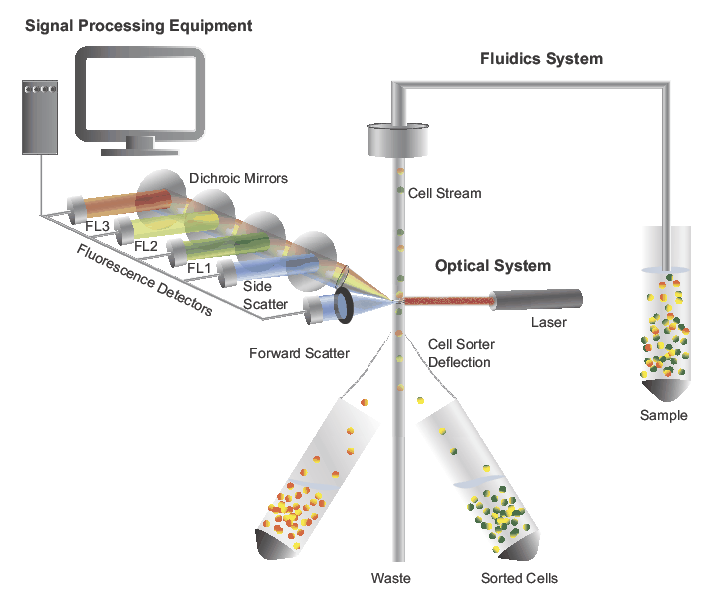
The panel includes surface markers to analyze activation (CD38, HLA-DR, ICOS/CD278), differentiation (CD45RA, CD27, CD28, CD57), expression of cytokine and chemokine receptors (CD25, CD127, CCR10, CCR6, CCR4, CXCR3, CXCR5, CRTH2/CD294), and co-inhibitory molecules and exhaustion (PD-1, CD223/LAG-3, TIGIT), which enables a deep characterization of PBMCs from peripheral blood. We have developed a 43 color panel to characterize most of the immune subsets within the peripheral immune system, including conventional T cells, unconventional T cells such as invariant natural killer T cells (iNKT), Gamma delta (γδ) T-cell subsets (TCR Vδ2, TCR Vγ9) and mucosal-associated invariant T cells (MAIT), B-cell subsets, natural killer (NK) cells, plasmacytoid dendritic cells, dendritic cell subsets, hematopoietic progenitor cells, basophils, and innate lymphoid cell (ILC) subsets (CD117, CRTH2). Spectral flowcytometry is capable of differentiating fluorochromes with significant overlap in the emission spectra, enabling the use of spectrally similar fluorochrome pairs such as Brilliant Blue 515 and FITC in a single panel. 
Although many flow cytometers can analyze 30–50 parameters, it is still challenging to develop a 40+ color panel for the phenotyping of immune cells using fluorochrome conjugated antibodies due to limitations in the availability of spectrally unique fluorochromes that can be excited by the commonly used laser lines (UV, Violet, Blue, Green/Yellow-green, and Red).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
